Introduction
Epsilon toxin (ETX) is produced by a plasmid (a small self-replicating strand of DNA within a bacterial cell) and is hosted by a bacterium called Clostridium perfringens (C. perfringens), which inhabits the intestine. C. perfringens is ubiquitous in nature and can be found as a normal component of decaying vegetation and animals, marine sediment, the intestinal tract of humans and other vertebrates, insects, and soil. It has the shortest reported generation time of any organism at 6.3 minutes in thioglycolate medium. C. perfringens (type A) is recognised as part of the human microbiome and is one of the most common causes of food poisoning in the United States. C. perfringens is a gram-positive, spore-forming, anaerobic bacillus that is widely present; its spores can survive long periods (decades) in hostile environments and will germinate when favourable conditions arise.
C. perfringens comprises five different types of bacteria (A to E), which cause various ailments, but it is only types B and D, which are not recognised as part of the human microbiome and which produce ETX. The plasmid in types B and D initially produces ETX as an inactive “prototoxin” in the gut, which is then activated by an enzyme (typically trypsin or chymotrypsin, which is prevalent in the gut). Once in its active state, the toxin can cross the intestine wall and thereby enter the bloodstream. To give an analogy, we could say that, if the prototoxin is represented as a “shirt and tie”, the trypsin rips off the “tie” to leave just the “shirt” as the active toxin.
Mechanism of Action
The diagrams below (reproduced from https://www.nature.com/articles/s41467-019-10645-8 and https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3649266 ).

The diagrams are reproduced from: “The pore structure of Clostridium perfringens epsilon toxin” (2109) Nature Communications (46) These show the toxin and how it morphs into a heptameric (seven sided) pore.
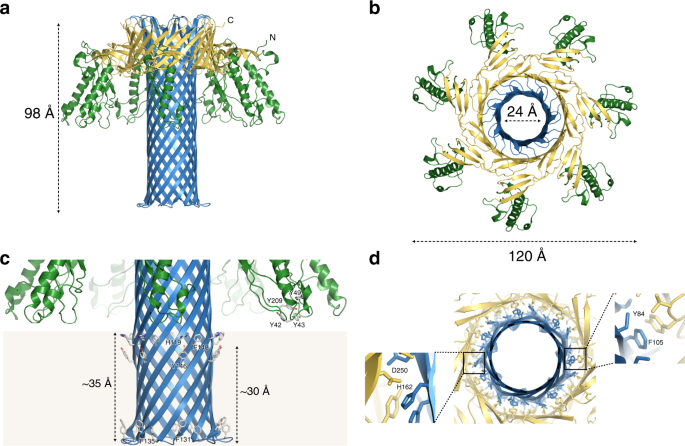

What does ETX do once in the Bloodstream?
ETX is known as a “pore-forming” toxin, whereby seven units of the toxin (“monomers”) bind together in certain cell walls of a host cell to form a heptameric (seven-sided) structure (“oligomer”). A good analogy would be that the monomers combine together in a cell wall to create a “funnel” between the cell and the blood vessel. This pore exposes the contents inside the cell to the outside, leading to rapid cell death (necrosis).
The following is an extract from an article by MR Popoff (2011) “Epsilon toxin: a fascinating pore‐forming toxin“ (14) describing the impact of ETX:
“The brain is the second organ, after the kidneys, where ETX accumulates massively. By contrast to kidneys, ETX binds to brain in an exclusively specific manner and with high affinity… This indicates that ETX passes the blood–brain barrier and recognizes specific cells or sites in the brain. Indeed, ETX has been shown to alter the integrity of the blood–brain barrier, permitting not only its own passage, but also that of macromolecules …. Such a rapid decrease in blood–brain barrier permeability facilitates rapid ETX accumulation in the brain …. Endothelial cells then show macroscopic alterations including swelling, abundance of clear vacuoles and loss of intracellular organelles, as well as protrusions or blebbing of the luminal surface. Later, their cytoplasm is very thin and the nuclei pyknotic, leading to a very attenuated capillary endothelium….”
ETX is therefore known to react with cells in the blood brain barrier (BBB), which are central nervous system (CNS) endothelial cells and, once in the brain, ETX also binds with high affinity to white matter – attacking myelin in oligodendrocytes, the optic nerve, spinal tissue as well as the kidney and Schwann cells. In addition to its pore forming ability, there is also evidence that ETX can damage oligodendrocytes without having to form pores. Oligodendrocytes lose potassium and take in sodium and calcium through osmosis and this leads to cell death.
Epsilon Toxin and Enterotoxaemia in Sheep and the possible link with Multiple Sclerosis
ETX is widely known to cause a highly lethal disease in sheep, called enterotoxaemia or pulpy kidney disease and it can also affect goats and cattle. The disease occurs in sheep when a bacterium, Clostridium perfringens (C. perfringens) (types B or D), which normally inhabits the animal’s intestines without causing problems, begins to multiply and produces ETX that poisons the animal and is often preceded by a sudden change to a low-fibre, high-carbohydrate diet. This can occur when sheep are moved onto lush, rapidly growing pasture or cereal crops, or when sheep are fed grain. Infected sheep can die within hours and ETX causes dysentery, severe oedema (the build-up of excess fluid) in the brain and kidney.
Although, for many years, the effects of disease from ETX have been extensively studied in animals, primarily ruminants, apart from one or two reported incidents, ETX was not previously associated with humans.
In the 1980s an Australian, Dr T G Murrell published an article, which put forward a hypothesis that there could be a link between sheep and MS (1). Dr Murrell instructed a laboratory to undertake ELISA testing of serum for antibodies to ETX from MS patients but this method, which is less sensitive than the Western Blotting technique used recently by K. Rumah et al and Exeter University (details below), did not provide any evidence of antibodies to ETX.
The first evidence of ETX in MS patients from Western Blotting was provided by Rumah et al (18) in 2013, with published results that showed 10% of MS patients were positive to ETX antibodies compared to 1% of controls. Rumah et al also isolated C. perfringens type B from a stool of a female patient, who had presented with the neurological symptoms of multiple sclerosis three months previously.
The Significance of ETX Breaching the Blood Brain Barrier
It is often thought that MS is an “auto-immune” disease and it does exhibit features, whereby host T effector cells and B cells attack the host’s myelin (myelin basic protein (MBP) and myelin oligodendrocyte glycoprotein (MOG)) in the brains of MS patients. However, Barnett and Prineas (2004) (5) found no or little evidence of lymphocytes in newly forming lesions when examined in MS patients, who died of other causes shortly after a MS relapse. This demonstrated that the first damage in a newly forming lesion is inflicted by something else other than host lymphocytes, which would be consistent with an infection from a toxin such as ETX.
It is known and accepted that MS in its early stages involves damage to oligodendrocytes within the brain (lesions), which first requires that the BBB must be breached. So, in order to identify the trigger of MS, we need to identify something, which is capable of firstly breaching the BBB. Whilst it is clear that there are “auto-immune” features of MS, we believe these are consequences or secondary effects from0 the initial insult being triggered by ETX.
The BBB is a highly selective semi-permeable border that separates the circulating blood from the brain and extracellular fluid in the CNS and originates from the endothelium during human embryonic development. The BBB is formed by endothelial cells of the capillary wall, astrocyte end-feet ensheathing the capillary, and pericytes embedded in the capillary basement membrane. This system allows the passage of water, some gases, and lipid-soluble molecules as well as the selective transport of molecules such as glucose and amino acids that are crucial to neural function.
The BBB restricts the diffusion of solutes in the blood (e.g. most bacteria) and large or hydrophilic molecules into the cerebrospinal fluid (CSF), while allowing the diffusion of hydrophobic molecules (O2, CO2, hormones) and small polar molecules. The BBB is therefore not easily breached – indeed significant medical research has been, and is being, undertaken to facilitate the uptake of drugs by the brain but so far with limited success. The BBB is breached, for instance, from a haemorrhagic stroke and certain bacteria such as meningeal bacteria. The other blood body barriers (including the blood periphery nervous system barrier) have different embryonic origins to the BBB and are not affected by ETX.
The main evidence for ETX causing MS symptoms comes from showing that the initial damage to the BBB, oligodentrocytes, blood retina barrier, optic nerve and lymphocytes can all be initiated by ETX in laboratory conditions. ETX, produced by C. perfringens types B and D is one of the most potent poisonous substances known. ETX binds to endothelial cells of brain capillary vessels before passing through the BBB.
References
The previous paragraphs have detailed that the presence of one protein, MAL, is associated with all the cells that are damaged by MS. There is now considerable evidence that MAL is necessary for binding and activity of ETX. References can be found as follows:
A) Rumah et al (23) published a paper in 2015 showing that MAL is required for binding and activity (cytotoxicity) of ETX.
B) Furthermore, Dr J Linden et al. (27) found evidence in 2015 that ETX specifically targets the myelin-forming cells of the CNS – mature oligodendrocytes – leading to cell death. She found ETX was highly specific for oligodendrocytes, as other cells of the CNS are unaffected and ETX-induced oligodendrocyte death results in demyelination and is dependent on the expression of MAL.
C) In a poster presentation, Dr J Linden also found that ETX binds to intestinal endothelial cells and confirmed ETX’s ability to bind to brain endothelial cells and cause BBB permeability. New evidence was provided that ETX binds to the retinal vasculature and causes blood retinal barrier (BRB) permeability. These observations are consistent with MS related retinal periphlebitis and macular oedema. In addition, ETX also binds to the blood vessels and myelin of the optic nerve, possibly explaining the high incidence of optic neuritis in MS patients.
D) A recent article published in July 2018 (39) provides evidence of a physical interaction between ETX and MAL, which was another missing piece of the jigsaw. A summary of the research follows:
“Epsilon toxin (Etx) from Clostridium perfringens is a pore-forming protein that crosses the Blood-Brain Barrier, binds to myelin and hence, has been suggested as a putative agent for the onset of multiple sclerosis, a demyelinating neuroinflammatory disease. Recently, Myelin and Lymphocyte protein (MAL) has been identified as a key protein in the cytotoxic effect of Etx, however the association of Etx with the immune system remains a central question. Here, we show that Etx selectively recognizes and kills only human cell lines expressing MAL through a direct Etx-MAL interaction. Experiments on lymphocytic cell lines reveal that MAL expressing T cells, but not B cells, are sensitive to Etx, and revealed the toxin as a molecular tool to distinguishing subpopulations of lymphocytes. The overall results open the door to investigate the role of Etx and Clostridium perfringens on inflammatory and autoimmune diseases like multiple sclerosis.”
E) A further experiment shows that MAL is critical for ETX to affect mice. There is a video of two mice, one a normal mouse given a lethal dose of ETX and the other a “knock-out MAL” transgenic mouse (i.e. one which has been bred without the MAL gene) and given 15 times the lethal dose of ETX. The knock-out MAL mouse survives while the normal mouse dies.
MAL is therefore known to be critical for ETX to form pores and damage cells. MAL is expressed in all the cell types that are damaged by MS. In conclusion, it is increasingly clear from the evidence that ETX can damage the exact cells, which are known to be damaged during a MS relapse and give rise to the early stage symptoms of MS.
